
Why Ascendo Biotechnology
First-in-class MoA targeting innate immune checkpoints
Addresses root cause of resistance, not just downstream effects

Strong potential for combination with PD-1 and other therapies

Broad applicability across oncology + autoimmune diseases
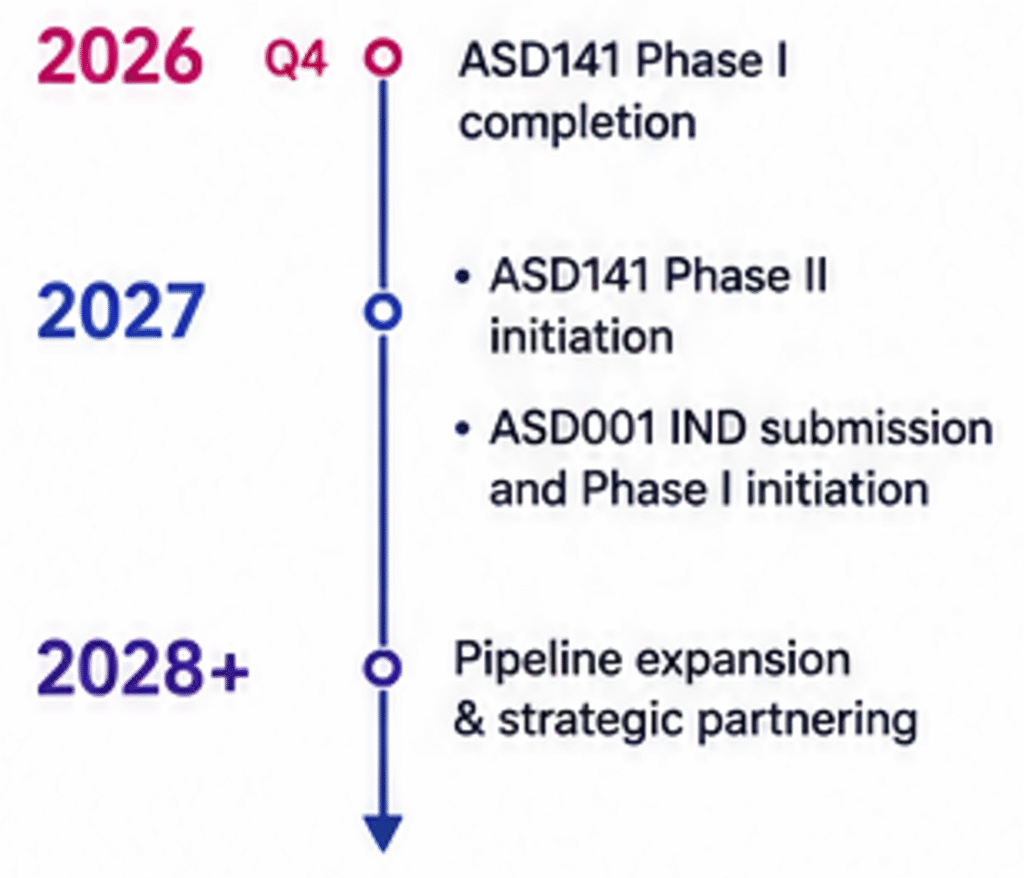
UPCOMING CATALYSTS
Multiple near-term de-risking milestones
- ASD141 Phase I completion (2026) -Phase II initiation (2027)
- ASD001 IND submission and Phase I initiation (2027)
- Pipeline expansion into next-generation innate immune programs
- Strategic partnering and licensing discussions
PARTNERING & INVESTMENT
Positioned to lead the next wave of immunotherapy beyond PD-1
Actively seeking:
- Co-development partners
- Out-licensing partners
- Strategic pharma partnerships
- Growth-stage investor

Partner of Choice for Immunotherapy Innovation
Ascendo Biotechnology is a partner of choice for novel immunotherapies targeting the interplay between innate and adaptive immunity.
We are actively seeking strategic partners to accelerate the development and commercialization of ASD141 and ASD001.
To explore partnership opportunities, please contact us.
